The U.S. Food and Drug Administration (FDA) is expected to propose a significant change to the labelling of packaged foods, requiring key nutritional information to be displayed on the front of the packaging in addition to the existing labels on the back. This move is aimed at making it easier for consumers to quickly access important dietary information, potentially encouraging healthier food choices.
The concept of front-of-package (FOP) nutrition labels is already in use in many countries. For example, Chile uses a stop sign symbol for items high in sugar, saturated fat, sodium, or calories, while Israel uses a red warning label. Singapore grades beverages based on their nutritional value.
Advocates have long pushed for the FDA to implement FOP labels, arguing that they help consumers make healthier choices and encourage manufacturers to improve their recipes. The FDA began seriously considering this change following the 2022 White House Conference on Hunger, Nutrition, and Health. Since then, the agency has reviewed research and conducted focus groups to explore label designs.
Among the designs tested by the FDA, some use traffic light colors to indicate levels of saturated fat, sodium, and added sugars, while others label products as “high in” certain nutrients, sometimes including the percentage of the recommended daily value.
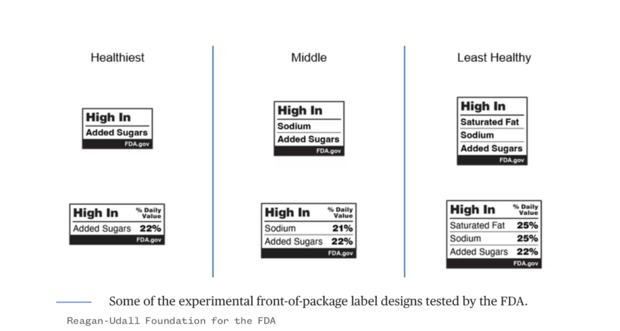
The FDA has yet to disclose the final design choice or the exact timing of the proposed rule, although it is expected this summer.
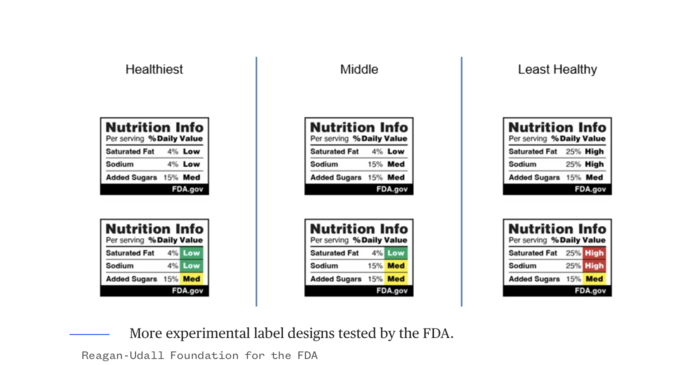
Trade associations such as the Consumer Brands Association and the FMI oppose mandatory interpretive labels, arguing they might cause unnecessary fear and do not provide meaningful context. These associations support the existing voluntary Facts up Front system, which highlights calories, saturated fat, sodium, and added sugars per serving, and can also include beneficial nutrients like potassium or fibre.

However, critics argue that voluntary systems do not have widespread use and lack the necessary context for consumers.
The FDA’s own review supports the effectiveness of FOP labels in helping consumers identify healthy foods, especially for those with lower nutrition knowledge and busy lifestyles.As obesity rates and diet-related diseases rise in the U.S., many believe that enhanced nutritional labeling could play a crucial role in improving public health.
Navigating Front-of-Package Nutrition Labels: Balancing Benefits and Challenges
Potential Benefits:
1. Increased Transparency:
o Consumers can easily see key nutritional details such as calories, sugars, fats, and sodium content without needing to look at the back of the package.
2. Improved Public Health:
o With nutritional information more prominently displayed, consumers may make healthier choices, potentially reducing rates of obesity, diabetes, and other diet-related diseases.
3. Consumer Awareness:
o Front-of-package labels can help raise awareness about the nutritional content of foods, leading to better-informed decisions and a greater understanding of what constitutes a healthy diet.
Resistance from Food Trade Associations: Despite the potential benefits, food trade associations are likely to resist the proposed changes for several reasons:
1. Increased Costs:
o Updating packaging to include new labels could be costly for food manufacturers, especially if they need to redesign their product packaging and distribution materials.
2. Market Impact:
o Products with less favorable nutritional profiles might see a decline in sales if consumers are deterred by the front-of-package information, affecting the bottom line of some companies.
3. Regulatory Burden:
o Additional labelling requirements may add to the regulatory burden on food manufacturers, requiring more compliance checks and adjustments to stay within the guidelines.
4. Consumer Confusion:
o There is a concern that too much information on the front of the package might overwhelm or confuse consumers, rather than helping them make better choices.
Considerations Moving Forward:
1. Design and Implementation:
o The design of the front-of-package labels needs to be clear, concise, and standardized to avoid confusion and ensure that the information is easily understandable.
2. Education and Outreach:
o The FDA might need to implement educational campaigns to help consumers understand the new labels and how to use them to make healthier food choices.
3. Industry Collaboration:
o Collaboration with food manufacturers and trade associations could help address their concerns and facilitate a smoother transition to the new labeling requirements.
4. Monitoring and Evaluation:
o Post-implementation, the impact of the new labels on consumer behavior and public health should be monitored to assess effectiveness and make any necessary adjustments.
The proposal for front-of-package nutritional labeling is part of a broader effort to enhance food labeling and promote public health, aligning with similar initiatives in other countries. The outcome of this proposal will depend on the FDA’s ability to balance the benefits to consumers with the concerns of the food industry.
For more information visit:
https://www.fda.gov/food/food-labeling-nutrition/front-package-nutrition-labeling

